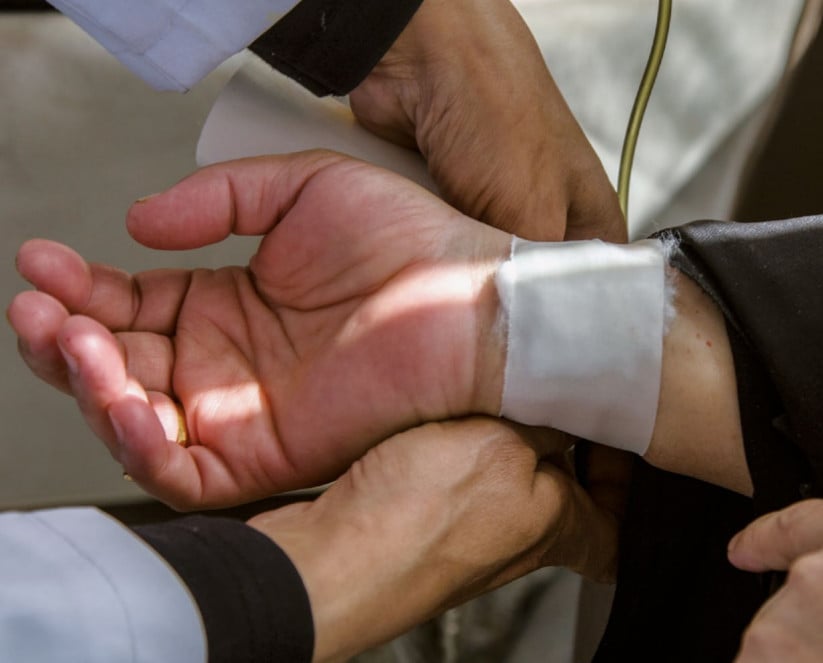
Egyptian patients targeted in clinical drugs trials from which they do not benefit
A new report reveals how foreign drug companies are targeting Egyptian cancer patients for trials of drugs from which they ultimately do not benefit.
Testing drugs has shifted in recent years from Europe and the United States to countries where trial costs are lower. Despite, or perhaps because of, the limited functioning of the legislative and monitoring power of the government since the Arab Spring in 2011, the number of clinical tests in Egypt has risen, making it the most popular country in Africa for testing drugs (after South Africa).
Industry-sponsored clinical drug trials in Egypt
The organizations Wemos, SOMO, Déclaration de Berne, Egyptian Initiative for Personal Rights and Shamseya for Innovative Community Healthcare Solutions commenced a joint study in 2015 on the situation of vulnerable testing subjects in Egypt. The report, Industry-sponsored clinical drug trials in Egypt: Ethical questions in a challenging context, published in June 2016, shows that international ethical standards are violated. Pharmaceutical companies do not always comply with Egyptian law (if present) and their own codes of conduct.
Cancer drugs, which account for more than half of all testing in Egypt, are a major concern. The study shows that subjects participate because they cannot get regular treatment. In Egypt, half of the population has no health insurance, resulting in 72% of all disease expenses being paid for out of pocket. The lack of access to treatment makes them vulnerable and unfit for a careful and well informed consent process, which lies at the heart of all ethical guidelines on clinical testing.
Walaa, a lung cancer patient and participant in a drug test says: “I was so happy to qualify for a treatment, I had lost all hope. Without reading I signed the consent form.”
Researchers also discovered at least three early stage trials (Phase I and Phase II) including investigational drugs that were not yet approved in Europe or the US. This is a clear contravention of Egyptian regulations, as Egypt forbids foreign drugs being tested on Egyptians first, to protect them from being used as guinea pigs.
Clinical trial treatment is often chosen because of the lack of access to proven and effective drugs
In Europe, cancer patients have access to treatment with proven efficacious and safe drugs. Only if this treatment does not work are patients offered the option of experimental medicine. Egyptian testing subjects, however, often have no choice other than the experimental treatment.
Annelies den Boer, global health advocate at Wemos, says: “When pharmaceutical companies test their drugs in countries where people have no access to health care they need to take additional measures to protect the safety and rights of subjects. They should not include cancer patients their experiments without first giving them the opportunity to undergo regular treatment.”
Drugs tested and approved are often not available on the Egyptian market
Another unethical issue is that Egyptians in many cases do not have access to drugs tested in their country – either because they are too expensive or because they have not been registered by the drugs company that tested them there. Of a sample of 24 medications tested in Egypt there was no marketing approval found for nine of them, while the same nine drugs are marketed in Europe or the US.
Irene Schipper, researcher at SOMO: “Not registering a drug in the country where it has been tested is both a violation of international ethical guidelines as well as a violation of the codes of conduct of individual pharmaceutical companies.” One in five drugs of the investigation costs more than 20 times the minimum monthly wage. This means that patients are ultimately unable to benefit from medications proven effective in tests carried out on them by the pharmaceutical industry.
Do you need more information?
-

Irene Schipper
Senior Researcher
Partners
Related content
-
Industry-sponsored clinical drug trials in Egypt Published on:
 Irene SchipperPosted in category:Publication
Irene SchipperPosted in category:Publication Irene Schipper
Irene Schipper
-
 Ethics and quality of drug testing in low income countries subject to additional pressure through outsourcingPosted in category:NewsPublished on:
Ethics and quality of drug testing in low income countries subject to additional pressure through outsourcingPosted in category:NewsPublished on: -
 Irene SchipperPosted in category:Publication
Irene SchipperPosted in category:Publication Irene Schipper
Irene Schipper -
 Experimental breast cancer drug unethically tested on Indian womenPosted in category:NewsPublished on:
Experimental breast cancer drug unethically tested on Indian womenPosted in category:NewsPublished on:

